Because the bond between the halogen and the carbon in the benzene ring aryl halide or a carbon participating in a double bond vinylic halide is much too strong stronger than that of an alkyl.
Sn2 vinyllic halide.
The student asked why do vinyl halides not do the sn2 reaction my answer was that two reasons exist for why the vinyl halide will not react with a nucleophile.
Classification allyic vinylic benzylic aryl halides.
They exhibit faster s n 2 reactivity than secondary alkyl halides because the bimolecular transition state is stabilized by hyperconjugation between the orbital of the nucleophile and the conjugated pi bond of the allylic.
There are many cases where allylic halides react preferentially by an mathrm s n 1 process.
For this reason alkenyl halides with the formula rch chx are sometimes called vinyl halides.
In organic chemistry a vinyl halide is a compound with the formula ch 2 chx x halide the term vinyl is often used to describe any alkenyl group.
From the perspective of applications the dominant member of this class of compounds is vinyl chloride which is produced on the scale of millions of.
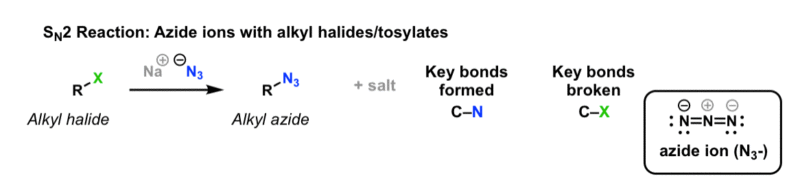
Allylic halides and tosylates are excellent electrophiles for bimolecular nucleophilic substitution reactions s n 2.
The substituents around a double bond are within the same plane therefore an s math n math 2 would give steric hindrance.
826 chapter 18 the chemistry of aryl halides vinylic halides and phenols.
A sn2 mechanism is not favoured for 3 reasons.
A sn1 sn2 mechanism on vinyl halide would look like this.
The substituents around a double bond are within the same plane therefore an sn2 would give steric hindrance as depicted in the above scheme.
Solvolysis of vinyl halides in very acidic media is an example.
A sn1 sn2 mechanism on vinyl halide would look like this.
Transition metal catalysis problem 18 2 arrange the following compounds according to increasing rate of elimination with naoc 2h 5 in c 2h 5oh what is the product in each case.
The picture below helps explain why this reaction is so much more difficult energetically more costly than the more common solvolysis of an alkyl halide.
Since both the allylic mathrm s n 1 and mathrm s n 2 reactions are stabilized there is a delicate balance between the two pathways.
The resultant vinylic carbocations are actually stable enough to be observed using nmr spectroscopy.
S n 2 reactions of allylic halides and tosylates.
A s math n math 2 mechanism is not favoured for 3 reasons.
Haloalkanes haroarenes part 1.
Why do allylic halides prefer sn2 reaction over sn1.
We can shift from one mechanism to the.
18 3 lack of reactivity of vinylic and aryl halides under s n1 conditions recall that tertiary and some secondary alkyl halides undergo.